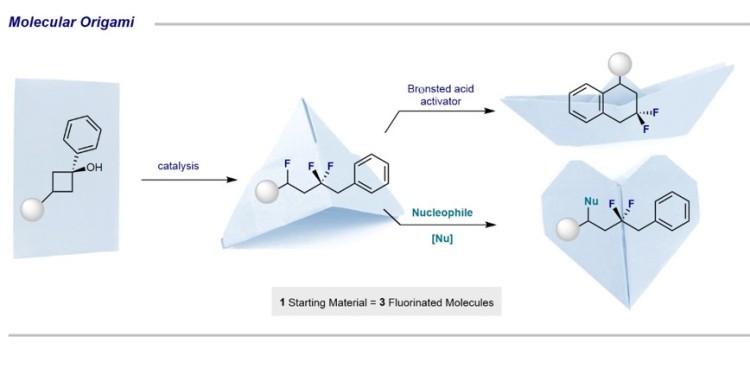
Chemists develop reaction cascade to produce fluorinated molecules
Fluorine is found rarely in naturally occurring organic molecules. However, this chemical element is indispensable for the production of pharmaceuticals or agrochemicals. Synthetic chemistry has an important role to play in the development of new fluorine-containing molecular fragments. Simple, modular synthesis strategies are extremely valuable. A team led by Prof. Ryan Gilmour at the Organic Chemistry Institute at the University of Münster have now developed a cascade reaction that enabled multiple fluorination reactions to occur through the sequential generation of reactive intermediates. Using inexpensive organic catalysts and simple starting materials, the group has shown that the substrate can be manipulated through a type of “molecular origami” to generate a new class of di- and trifluorinated molecules in a single operation. The study is published in the journal "Nature Communications".
The step-wise construction of complex fluorinated molecules may require multiple purification steps. This has implications in terms of costs, time-management and the generation of waste. Dr Joel Häfliger and Dr Louise Ruyet from Ryan Gilmour's research group discovered that by fine-tuning the reaction conditions, multiple sequential reactions are possible in a one pot fashion. In this way, they generated three new classes of complex fluorinated products from simple cyclobutanol derivatives. "The strategy has parallels with the art of origami, where you fold complex figures from a simple piece of paper," Louise Ruyet illustrates. "This principle can be transferred to our chemical method."

This work was funded by the European Research Council (ERC Consolidator Grant RECON “Reprogramming Conformation by Fluorination: Exploring New Areas of Chemical Space”. Grant agreement ID: 818949).
Original publication
J. Häfliger, L. Ruyet, N. Stübke, C. G. Daniliuc and R. Gilmour (2023): Integrating I(I)/I(III) Catalysis in Reaction Cascade Design Enables the Synthesis of gem-Difluorinated Tetralins from Cyclobutanols. Nature Communications, 14, 3207; DOI: 10.1038/s41467-023-38957-w
